|
The Consortium for Advanced Practice Providers offers programmatic accreditation for postgraduate nurse practitioner and joint nurse practitioner and physician associate training programs, designed by NP and PA postgraduate program directors, for NP and PA postgraduate training directors. The Consortium exists to support new and ongoing postgraduate training programs for nurse practitioners and physician associates in the achievement and maintenance of the highest standards of rigor and quality, consistent with achieving the goal of an expert healthcare workforce prepared to meet the needs of patients and the society as a whole. |

Timeline and Process
The following information outlines the process and provides an estimate of time to complete each step. In general, the Accreditation review takes eight to twenty-four months with appropriate time and resources.
- Click here to see the Accreditation Fact Sheet
- Click here to see a slide show presentation giving an overview of Consortium’s Accreditation Process
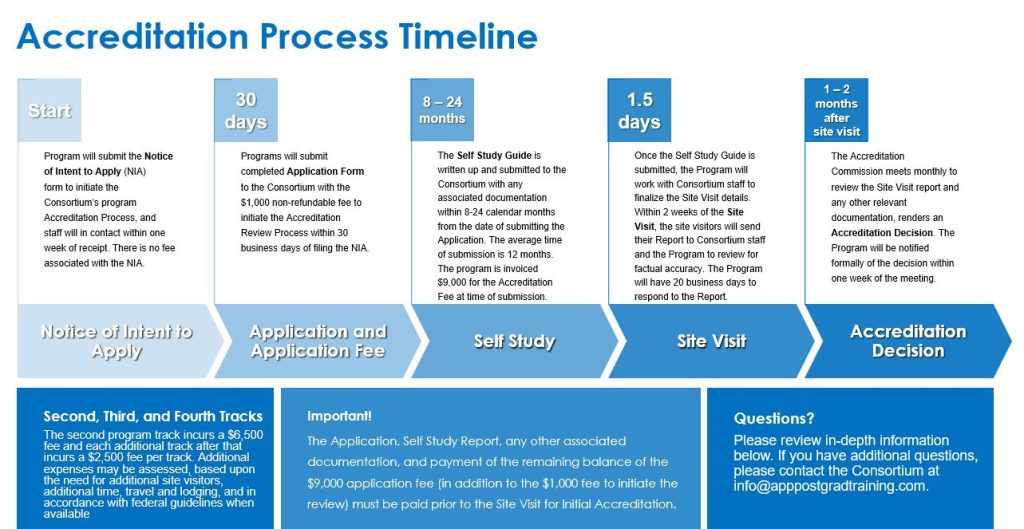
Application Process:
Program will submit the Notice of Intent to Apply (NIA) form to initiate the Consortium’s program Accreditation Process. This allows the Consortium to plan resource allocation and schedule for the coming months. There is no fee associated with the NIA. Click the APPLY tab in the upper right hand corner of the screen to access the Notice of Intent to Apply and the Application forms.
Consortium for Advanced Practice Providers staff will contact the program’s designated contact person within one week of receipt of Notice of Intent to Apply, will open a program-specific Accreditation file, and will notify the Accreditation Commission of receipt. Notice of the Consortium’s receipt of the NIA will be sent electronically to the program, with a request to confirm receipt.
The Consortium will establish a due date for submission of the completed Application of 30 business days from receipt of Notice of Intent to Apply. (Please note: other documentation will be required during subsequent steps in the accreditation review process). The Consortium will also offer an opportunity for a virtual meeting to answer any questions about the Accreditation Process during that 30 day period. If the Application, including the $1,000 fee, is not received by the established date, the Consortium will notify the program that the Notice of Intent to Apply will be considered inactive and no further action will be taken. Programs may notify the Consortium that they would like to reactivate their NIA at any time. Reactivation will require an updated NIA.
Programs will submit completed Application form to the Consortium with the $1,000 non-refundable fee to initiate the Accreditation Review Process within 30 business days of filing the NIA. The Consortium staff will review the Application for completion. If incomplete, the program will be contacted and the Application will be returned for completion. If complete, Consortium will notify the program that Application is complete. A tentative site visit date will be set for approximately eight to twenty-four months from the date of receipt of the Application. The Application will be forwarded to the Accreditation Commission for review. Consortium staff will serve as the intermediary between the Program and the Accreditation Commission.
Consortium staff will schedule an initial call with the program within 30 business days of receipt of Application. Consortium staff will also be available for regularly scheduled technical assistance calls throughout the review process. Programs are encouraged to contact the Consortium at any time with questions regarding the Self Study Guide, the Accreditation Standards, as well as the process for Initial Accreditation and for Renewal of Accreditation. Further, programs are welcome to submit sections of the required documentation throughout the process. Programs will notify the Consortium when all required documentation has been submitted. Applications not completed will be kept on file for one year and must be updated when submitted.
Self Study:
The purpose of the Self Study is to conduct an internal evaluation of the program’s adherence to the Consortium for Advanced Practice Providers’ Accreditation Standards. The program conducts their Self Study. Tools to assist in the Self Study process include the Accreditation Program Self-Assessment and the Site Visit Agenda for the Accreditation Site Visit. The findings of the Self Study are written up and submitted to the Consortium with any associated documentation within eight to twenty-four calendar months from the date of submitting the Application. The average time of submission is 12 months.
The Application, Self Study Report, any other associated documentation, and payment of the remaining balance of the $10,000 application fee (in addition to the $1,000 fee to initiate the review) for Initial Accreditation and the $4,500 review fee for Renewal of Accreditation, must be received prior to further accreditation review action, such as conducting the Site Visit.
The second program track incurs a $6,500 fee and each additional track after that incurs a $2,500 fee per track. Additional expenses may be assessed, based upon the need for additional site visitors, additional time, travel and lodging, and in accordance with federal guidelines when available.
For a copy of the Self Study Guide in a Google Doc Format, please use this link to make a copy to your Google Drive. For a copy of the Self Study Guide in Microsoft Word format, please use this link to download a copy.
Site Visit:
Prior to the Site Visit, the Accreditation Commission will review all materials, assign and prepare the site visitor(s). Consortium staff will coordinate the collaborative process between the Accreditation Commission, the program of finalizing the site visit dates, and developing the agenda. The staff will serve as the point of contact for the program and will keep open lines of communication with the Accreditation Commission and the program. The Consortium staff will keep the program updated and inform the program of any major areas of concern that the program needs to address prior to the Site Visit.
The Consortium makes arrangements for the site visitors’ accommodations and transportation. There will be open communication through the process between the Consortium staff and the Program Director. Activities during the 1.5 day On-Site Visit include (but are not limited to): meeting with executive and programmatic leadership; observing clinical and didactic sessions; meeting with preceptors, trainees and alumni; reviewing documentation including credentialing files; public commentary submitted via the Consortium website; drafting of Site Visit Report and conducting an final exit meeting when site visit team shares observations with program representatives. After the site visit, the site visitors submit their report to Consortium staff, who then forward it to the program for review of factual accuracy. Following the program’s review, the report is then submitted to the Accreditation Commission.
Accreditation Decision:
Accreditation Commission meets to review Site Visit report and any other relevant documentation, renders an Accreditation Decision. The Program will be notified formally of the decision within one week of the decision. The Certificate of Accreditation will be issued within one week of the Accreditation Award notification. Accredited programs will be given access to the Consortium for Advanced Practice Providers’ Accreditation Seal for use in the organization’s communications. Accreditation is for a period of up to three (3) years for Initial Accreditation and for up to five (5) years for Renewal of Accreditation. The Action will be posted on the Consortium’s website.
Categories of Accreditation:
Initial Accreditation: awarded to a program that is in full compliance with the Standards and has not been previously accredited by Consortium in the immediately preceding time period.
Deferral of Accreditation: Under unusual circumstances, the Accreditation Commission may elect to defer a pending accreditation decision
Voluntary Withdrawal of Accreditation: The program elects to formally withdraw from accreditation status, at any time, for any reason. The program may reapply for Initial Accreditation at any time in the future, without prejudice
Denial of accreditation: The program demonstrates persistent and substantial non-compliance with the standards. This is a reportable adverse action
Rescinding of Accreditation: With persistent noncompliance with the accreditation standards or upon the discovery of an egregious violation of the standards, the Accreditation Commission may rescind a program’s accreditation. This is a reportable adverse action.
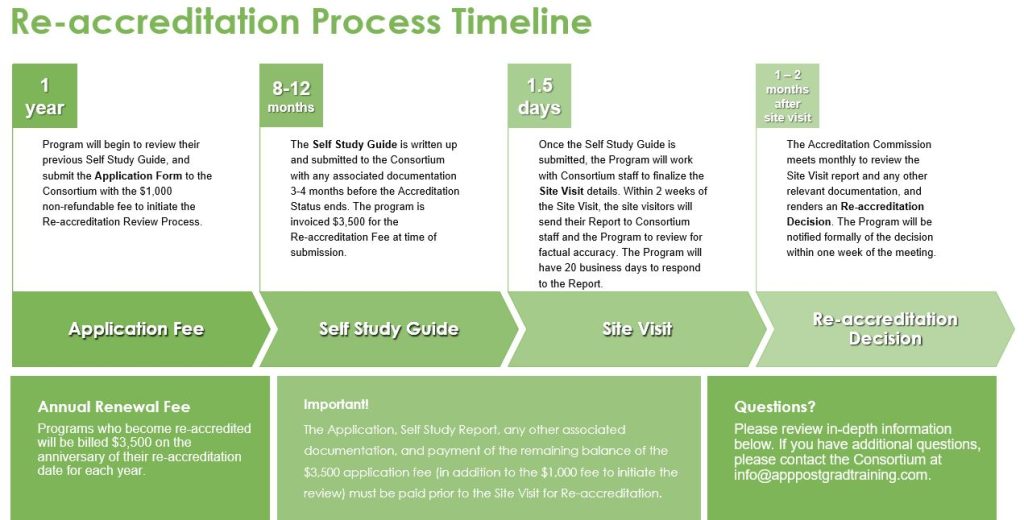
Re-accreditation Process:
Program will submit the Application form to initiate the Consortium’s Re-accreditation Process. This allows the Consortium to plan resource allocation and schedule for the coming months. Click the APPLY tab in the upper right hand corner of the screen to access the Application form.
Programs should start to consider the Re-accreditation Process six to twelve months before the Accreditation period lapses. The Program should plan to submit their Self Study Guide no later than three to four months prior to the Accreditation period end date. The Site Visit should be planned for two to three months prior to the Accreditation period end date. The Re-accreditation decision will be made one month before the Accreditation period end date. The Consortium will offer an opportunity for a virtual meeting to answer any questions about the Re-accreditation Process.
Programs will submit completed Application form to the Consortium with the $1,000 non-refundable fee to initiate the Re-accreditation Review Process. The Consortium staff will review the Application for completion. If incomplete, the program will be contacted and the Application will be returned for completion. If complete, Consortium will notify the program that Application is complete. A tentative site visit date will be set from the date of receipt of the Application. The Application will be forwarded to the Accreditation Commission for review. Consortium staff will serve as the intermediary between the Program and the Accreditation Commission.
Consortium staff will also be available for regularly scheduled technical assistance calls throughout the re-accreditation process. Programs are encouraged to contact the Consortium at any time with questions regarding the Self Study Guide and the Accreditation Standards. Further, programs are welcome to submit sections of the required documentation throughout the process. Programs will notify the Consortium when all required documentation has been submitted.
Self Study:
The purpose of the Self Study is to conduct an internal evaluation of the program’s adherence to the Consortium for Advanced Practice Providers’ Accreditation Standards. The program conducts their Self Study. Tools to assist in the Self Study process include the Accreditation Program Self-Assessment and the Site Visit Agenda for the Accreditation Site Visit.
The Application, Self Study Report, any other associated documentation, and payment of the remaining balance of the $4,500 re-accreditation fee must be received prior to further accreditation review action, such as conducting the Site Visit.
Site Visit:
Prior to the Site Visit, the Accreditation Commission will review all materials, assign and prepare the site visitor(s). Consortium staff will coordinate the collaborative process between the Accreditation Commission, the program of finalizing the site visit dates, and developing the agenda. The staff will serve as the point of contact for the program and will keep open lines of communication with the Accreditation Commission and the program. The Consortium staff will keep the program updated and inform the program of any major areas of concern that the program needs to address prior to the Site Visit.
The Consortium makes arrangements for the site visitors’ accommodations and transportation. There will be open communication through the process between the Consortium staff and the Program Director. Activities during the 1.5 day On-Site Visit include (but are not limited to): meeting with executive and programmatic leadership; observing clinical and didactic sessions; meeting with preceptors, trainees and alumni; reviewing documentation including credentialing files; public commentary submitted via the Consortium website; drafting of Site Visit Report and conducting an final exit meeting when site visit team shares observations with program representatives. After the site visit, the site visitors submit their report to Consortium staff, who then forward it to the program for review of factual accuracy. Following the program’s review, the report is then submitted to the Accreditation Commission.
Accreditation Decision:
Accreditation Commission meets to review Site Visit report and any other relevant documentation, renders an Accreditation Decision. The Program will be notified formally of the decision within one week of the decision. The Certificate of Accreditation will be issued within one week of the Re-accreditation Award notification. Re-accredited programs will be given access to the Consortium for Advanced Practice Providers’ Accreditation Seal for use in the organization’s communications. Re-accreditation is for a period of up to five (5) years for Renewal of Accreditation. The Action will be posted on the Consortium’s website.
Updated and Approved on 07/10/2024

